SolutionHard
Question
The fraction of phenol dimerised in benzene if 20 g of phenol in 1 kg benzene exhibits a freezing point depression of 0.69 K. (Kf benzene = 5.12 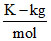 ), (MW phenol = 94)
), (MW phenol = 94)
Options
A.0.74
B.0.37
C.0.46
D.0.64
Solution
ᐃTf = i. kb.m
0.69 = i ×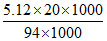
i = 0.63
for dimerization i = 1 - α/2
0.63 = 1 - α/2
α = 0.74.
0.69 = i ×
i = 0.63
for dimerization i = 1 - α/2
0.63 = 1 - α/2
α = 0.74.
Create a free account to view solution
View Solution FreeMore Solution Questions
Y gm of non-volatile organic substance of molecular mass M is dissolved in 250 gm benzene. Molal elevation constant of b...The normal freezing point of nitrobenzene is 278.82 K. A non-volatile solute is dissolved in it and a solution of molali...Aqueous solution of mixture of Ni2+, Ag+, Pb2+, Ca2+, and Al3+ is treated with dil.HCl and H2S, ppt.obtained is removed ...Which of the following liquid pairs shows a positive deviation from Raoult′s law ?...1 mol each of 1the following compounds is dissolved in 1L of solution. Which will have the largest ᐃTb value ?...