SolutionHard
Question
Observe the P-T phase diagram for a given substance A. Then melting point of A(s), boiling point of A(l), critical point of A and triple point of A (at their respective pressures) are respectively :
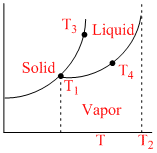

Options
A.T1, T2, T3, T4
B.T4, T3, T1, T2
C.T3, T4, T2, T1
D.T2, T1, T3, T4
Solution
T1 = Tripal point temp, T2 = Critical point temp., T3 = M.P.
T4 = B.P.
T4 = B.P.
Create a free account to view solution
View Solution FreeMore Solution Questions
Ethylene glycol is used as an antifreeze in a cold climate. Mass of ethylene glycol which should be added to 4 kg of wat...Which statement comparing solutions with pure solvent is not correct...Which of the following is/are correct for an ideal binary solution of two volatile liquids (eg. benzene & toluene)?...18 g of glucose (C6H12O6) is added to 178.2 g of water. The vapour pressure of water for this aqueous solution at 100oC ...PH3 (Phosphine) when passed in aqueous solution of CuSO4 it produce -...