SolutionHard
Question
Available are 1L of 0.1 M NaCl and 2L of 0.2 M CaCl2 solutions. Using only these two solutions what maximum volume of a solution can be prepared having [Cl-] = 0.34 M exactly. Both electrolytes are strong
Options
A.2.5 L
B.2.4 L
C.2.3 L
D.None of these
Solution
Let volumes takne by ′x′ & ′y′ litres, so 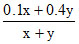 = 0.34 & Vg = (x + y) (to be maximised)
= 0.34 & Vg = (x + y) (to be maximised)
so y = 4x so for maximum volume
y = 2L & x = 1/2L
so y = 4x so for maximum volume
y = 2L & x = 1/2L
Create a free account to view solution
View Solution FreeMore Solution Questions
For a solution of 0.849 g of mercurous chloride in 50 g of HgCl2(l) the freezing point depression is 1.24oC. Kf for HgCl...Vapour pressure of CCl4 at 25oC is 143 mmHg. 0.5 g of a non-voltaile solute (molar mass = 65 mol-1) is dissolved in 100 ...Moles of K2SO4 to be dissolved in 12 mol water to lower its vapour pressure by 10 mmHg at a temperature at which vapour ...Benzene and naphthalene form an ideal solution at room temperature. For this process, the true statement(s)is(are)...For the given electrolyte AxBy, the degree of dissociation ′a′ can be given as...