SolutionHard
Question
Moles of K2SO4 to be dissolved in 12 mol water to lower its vapour pressure by 10 mmHg at a temperature at which vapour pressure of pure water is 50 mm is :
Options
A.3 mol
B.2 mol
C.1 mol
D.0.5 mol
Solution
Lowering of V.P. is colligative property
thus, iK2SO4 = 1 + (y - 1)x = 1 + 2x = 3
∴ If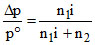
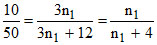
n1 = 1
thus, iK2SO4 = 1 + (y - 1)x = 1 + 2x = 3
∴ If
n1 = 1
Create a free account to view solution
View Solution FreeMore Solution Questions
The osmotic pressure of a solution may be increased by...Temperature does not affect :...The boiling point of a 2% (w/w) aqueous solution of a non-volatile and non-electrolyte solute is 0.102°C higher than tha...A liquid is in equilibrium with its vapour at its boiling point. On average, the molecules in the two phases have equal...Which has maximum osmotic pressure at temperature T :...