SolutionHard
Question
For the given electrolyte AxBy, the degree of dissociation ′a′ can be given as
Options
A.α = 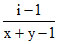
B.i = (1 - α) + xα + yα
C.α = 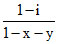
D.None
Solution
AxBy ⇋ xAm+ + yBn+
Initial moles n o o
At eq b. n(1-α) nxα nyα
i =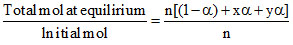
i = (1- α) + xα + yα
It can also seen that all other expressions imply the same thing.
(A) α =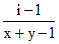 (B) i = (1 - α) + xα + yα. (C)
(B) i = (1 - α) + xα + yα. (C) 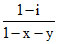
Initial moles n o o
At eq b. n(1-α) nxα nyα
i =
i = (1- α) + xα + yα
It can also seen that all other expressions imply the same thing.
(A) α =
Create a free account to view solution
View Solution FreeMore Solution Questions
Under the condition of similar temperature, which of the following solution will have minimum vapour pressure?...In KI solution, mercuric iodide is added. The osmotic pressure of resultant solution will...Consider the following aqueous solutions.I. 2.2 g Glucose in 125 mL of solution.II. 1.9 g Calcium chloride in 250 mL of ...CuSO4(aq) + NaOH(aq) → Pale blue ppt Intense blue colour Pale blue solutionThe pale blue solution is :-...The solubility of N2(g) in water exposed to the atmosphere, when the partial pressure is 593 mm is 5.3 × 10-4 M. It...