SolutionHard
Question
Vapour pressure of CCl4 at 25oC is 143 mmHg. 0.5 g of a non-voltaile solute (molar mass = 65 mol-1) is dissolved in 100 mL of CCl4 (density = 1.538 g mL-1). Vapour pressure of solution is
Options
A.141.9 mmHg
B.94.4 mmHg
C.99.3 mmHg
D.144.1 mmhg
Solution
Wt .of CCl4 = 1.538 × 100 = 153.8 gm
so moles of CCl4 =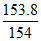 ≈ 1
≈ 1
moles of solute =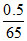 = 0.00769
= 0.00769
Now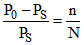
or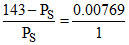
PS = 141.9 mm Hg.
so moles of CCl4 =
moles of solute =
Now
or
PS = 141.9 mm Hg.
Create a free account to view solution
View Solution FreeMore Solution Questions
A complex of iron and cyanide ions is 100% ionised at 1m (molal). If its elevation in b.p. is 2.08. Then the complex is ...Kf for water is 1.86 K kg mol-1. If your automobile radiator holds 1.0 kg of water, how many grams of ethylene glycol (C...Which aqueous solution can not be stirred with silver spoon :-...Which is a colligative property?...Which one is not equal to zero for an ideal solution:-...