SolutionHard
Question
For a solution of 0.849 g of mercurous chloride in 50 g of HgCl2(l) the freezing point depression is 1.24oC. Kf for HgCl2 is 34.3. What is the state of mercurous chloride in HgCl2 ? (Hg - 200, Cl - 35.5)
Options
A.as Hg2Cl2 molecules
B.as HgCl molecules
C.as Hg+ and Cl- ions
D.as Hg22+ and Cl- ions
Solution
1.24 = 34.3 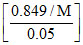 ⇒ M = 469.68
⇒ M = 469.68
∴ as Hg2Cl2 molecules.
∴ as Hg2Cl2 molecules.
Create a free account to view solution
View Solution FreeMore Solution Questions
A graph plotted between vs , d (where P is osmotic pressure of solution of a solute of mol. wt. m and d is its density a...Which of the following aqueous solution will have the highest boiling point?...When glycerin is added to a litre of water, then which of the following is observed?...Which combination of (I) vapour pressure, (II) intermolecular forces and (III) ᐃHvap (latent heat of vaporisation)...Which aqueous solution can not be stirred with silver spoon :-...