SolutionHard
Question
Aqueous solution of barium phosphate which is 100% ionised has ᐃTf / Kf as 0.05. Hence, given solution is
Options
A.0.01 molal
B.0.02 molal
C.0.04 molal
D.0.05 molal
Solution
Ba3(PO4)2 ⇋ 3 Ba2+ + 2 PO43-
value of i = 5 (100% ionised)
so ᐃTf = i kf m
so m =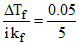 = 0.01
= 0.01
value of i = 5 (100% ionised)
so ᐃTf = i kf m
so m =
Create a free account to view solution
View Solution FreeMore Solution Questions
Which of the following combinations are correct for a binary solution, in which the solute as well as solvent are liquid...The total concentration of dissolved particles inside red blood cells is approximately 0.30 M and the membrane surroundi...A Solution of sucrose (molar mass = 342 gm-mol-1) has been prepared by dissolving 68.5 g of sucrose in 1000 g of water. ...3% aqueous solution of dentrose (mol wt = 180) is isotonic with 2% aqueous solution of another covalent solute in water ...How many grams of sucrose (molecular weight = 342) should be dissolved in 100 g water in order to produce a solution wit...