SolutionHard
Question
Which of the following combinations are correct for a binary solution, in which the solute as well as solvent are liquid ?
Options
A.C6H6 and C6H5CH3; ᐃsolH > 0; ᐃsolV = 0
B.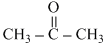 and CHCl3; ᐃsolH < 0; ᐃsolV < 0
and CHCl3; ᐃsolH < 0; ᐃsolV < 0
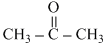 and CHCl3; ᐃsolH < 0; ᐃsolV < 0
and CHCl3; ᐃsolH < 0; ᐃsolV < 0C.H2O and HCl; ᐃsolH > 0; ᐃsolV < 0
D.H2O and CH2OH; ᐃsolH > 0; ᐃsolV < 0
More Solution Questions
The average osmotic pressure of human blood is 7.8 bar t 37oC. What is the concentration of an aqueous NaCl solution tha...If relative decrease in vapour pressure is 0.4 for a solution containing 1 mol NaCl in 3 mol H2O, NaCl is .... % ionised...When H2S gas is passed through the HCl containing aqueous solution of CuCl2, HgCl2, BiCl3, and CoCl2, Which does not pre...Consider the following reactions.$${PbCl}_{2} + K_{2}{CrO}_{4} \rightarrow \text{ }A + 2KCl $$(Hot solution)$${A + NaOH ...The only incorrect statement regarding Henry’s law for a gas dissolving in water is...