SolutionHard
Question
3% aqueous solution of dentrose (mol wt = 180) is isotonic with 2% aqueous solution of another covalent solute in water at 25oC. The molar mass of the solute is :-
Options
A.60
B.120
C.180
D.90
Solution
For isotonic
Dextrose = covalent
π1 = π2
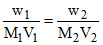
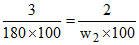
Dextrose = covalent
π1 = π2
Create a free account to view solution
View Solution FreeMore Solution Questions
Mole fraction of C3H5(OH)3 in a solution of 36 g of water and 46 g of glycerine is :...On mixing 3g of non-volatile solute in 200 mL of water, its Boiling point (100oC) be comes 100.52oC. If Kb for water is ...The osmotic pressure of equimolar solutions of BaCl2, NaCl and glucose will be in the order...300 ml, N/100 HNO3 solution is mixed with 200 ml, N/100 KOH solution. The [H+] in the resultant solution is :-...An aqueous solution of hydrochloric acid...