SolutionHard
Question
The vapour pressure of pure liquid solvent A is 0.80 atm. When a non-volatile substance B is added to the solvent, its vapour pressure drops to 0.60 atm. Mole fraction of the component B in the solution is :
Options
A.0.50
B.0.25
C.0.75
D.0.40
Solution
Acc. to R.L. V.P. ⇒ 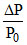 = XB
= XB
XB =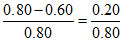 = 0.25.
= 0.25.
XB =
Create a free account to view solution
View Solution FreeMore Solution Questions
Density of 1M solution of a non-electrolyte C6H12O6 is 1.18 g/mL. If Kf (H2O) is 1.86o mol-1 kg, solution freezes at :...Insulin (C2H10O5)n is dissolved in a suitable solvent and the osmotic pressure p of the solution of various concentratio...Liquids A and B form an ideal solution. The plot of $\frac{1}{X_{A}}$ (Y-axis) versus$\frac{1}{Y_{A}}$ (X-axis) (where X...A liquid is in equilibrium with its vapours at its boiling point. On the average the molecules in the two phases have eq...Freezing point of an aqueous solution is (-0.186)oC. Elevation of boiling point of the same solution is Kb = 0.512 0C, K...