SolutionHard
Question
Freezing point of an aqueous solution is (-0.186)oC. Elevation of boiling point of the same solution is Kb = 0.512 0C, Kf = 1.86oC, find the increase in boiling point.
Options
A.0.186oC
B.0.0512oC
C.0.092oC
D.0.2372oC
Solution
ᐃTb = Kb × 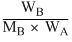 × 1000 ; ᐃTf
× 1000 ; ᐃTf 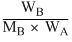 × 1000 ;
× 1000 ; 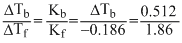 = 0.0512oC
= 0.0512oC
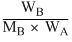 × 1000 ; ᐃTf
× 1000 ; ᐃTf  × 1000 ;
× 1000 ; 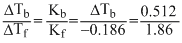 = 0.0512oC
= 0.0512oCCreate a free account to view solution
View Solution FreeMore Solution Questions
If relative decrease in vapour pressure is 0.4 for a solution containing 1 mol NaCl in 3 mol H2O, NaCl is .... % ionised...Which of the following solution will have pH = 13 ?...FeCl3 solution (side X) on reaction with K4[Fe(CN)6] (side Y) in aqueous solution gives blue colour. These are separated...What is molarity of HCl in a solution prepared by dissolving 5.5g HCl in 200 g ethanol. If the density of the solution i...An aqueous solution of a solute AB has b.p. of 101.08oC (AB is 100% ionised at boiling point of the solution) and freeze...