SolutionHard
Question
Insulin (C2H10O5)n is dissolved in a suitable solvent and the osmotic pressure p of the solution of various concentration (in kg/m3) is measured at 20oC. The slope of a plot of p against c is found to be 8.134 × 10-3 (SI units) The molecular weight of the insulin (in kg/mol) is :
Options
A.4.8 × 105
B.9 × 105
C.293 × 103
D.8.314 × 105
Solution
π = CRT
π =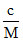 RT C = moles/liter, c = kg/m3
RT C = moles/liter, c = kg/m3
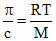
M =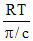 [π/c = 8.314 × 10-3]
[π/c = 8.314 × 10-3]
[T = 293 k]
M =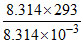 = 293 × 103
= 293 × 103
π =
M =
[T = 293 k]
M =
Create a free account to view solution
View Solution FreeMore Solution Questions
Which of the following liquid pairs do not show positive deviation from Raoult’s law?...Henry’s law constant for CO2 in water is 1.6 × 108 Pa at 298 K. The quantity of CO2 in 500 g of soda water when packed u...Maximum osmotic pressure will be shown by which of the following solution (all 90% ionized)?a) 0.1 M-AlCl3 (b) 0.1 M-BaC...Select correct statement ?...At $T(K),2$ moles of liquid A and 3 moles of liquid B are mixed. The vapour pressure of ideal solution formed is 320 mm ...