Atomic StructureHard
Question
The electron in the hydrogen atom jumps from excited state (n = 3) to its ground state (n = 1) and the photons thus emitted irradiate a phonsenstive material. If the work function of material is 5.1 eV, the stopping potential is estimated to be (the energy of the electron in nth state En = - 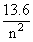 eV)
eV)
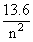 eV)
eV)Options
A.5.1 V
B.12.1 V
C.17.2 V
D.7 V
Solution
Energy released when electrons in the atom jumps from excited state (n = 3) to ground state (n = 1) is
E = h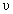 = E3 - E1 =
= E3 - E1 = 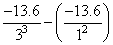
=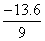 + 13.6 = 12.1eV
+ 13.6 = 12.1eV
Therefore, stopping potential
eV0 = hv - φ0
= 12.1 - 5.1 [∵ work function φ0 = 5.1]
V0 = 7V.
E = h
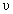 = E3 - E1 =
= E3 - E1 = 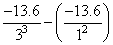
=
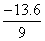 + 13.6 = 12.1eV
+ 13.6 = 12.1eVTherefore, stopping potential
eV0 = hv - φ0
= 12.1 - 5.1 [∵ work function φ0 = 5.1]
V0 = 7V.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Two insulating plates are both uniformly charged in such a way that the potential difference between them is V2 - V1 = 2...Nuclear reactions are given as -(i) ⎕ (n, p)15p32(ii) ⎕ (p, α)8O16(iii) 7N14 (⎕, p) 6C14 missing ...Binding energy per nucleon versus mass number curve for nuclei is shown in figure. W, X, Y and Z are four nuclei indicat...Let Mp be the mass of photon, mn the mass of neutron. M1 the mass of necleus and M2 the mass of nucleus. Then...Hydrogen (1H1), Deuterium (1H2) singly ionised Helium (2He4)+ and doubly ionised lithium (3Li6)++; all have one electron...