Atomic StructureHard
Question
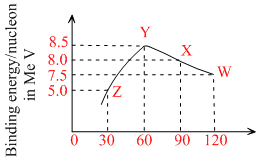
Binding energy per nucleon versus mass number curve for nuclei is shown in figure. W, X, Y and Z are four nuclei indicated on the curve. The process that would release energy is

Options
A.Y → 2Z
B.W → X + Z
C.W → 2Y
D.X → Y + Z
Solution
Energy is released in a process when total binding energy of nucleus (= binding energy per nucleon × number of nucleons) is increased or we can say, when total binding energy of products is more than the reactants. By calculation we can see that only in option (c), this happens.
Given : W → 2Y
Binding ebergy of reactant = 120 × 7.5 = 900 MeV
and binding energy of products = 2 (60 × 8.5)
= 1020 MeV > 900 MeV.
Given : W → 2Y
Binding ebergy of reactant = 120 × 7.5 = 900 MeV
and binding energy of products = 2 (60 × 8.5)
= 1020 MeV > 900 MeV.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
238U has 92 protons and 238 nucleons. It decays by emitting an Alpha particle and becomes...The ground state energy of hydrogen atom is -13.6eV. What is the potential energy of the electron in this state?...If 13.6 eV energy is required to ionize the hydrogen atom, then the energy required to remove an electron from n = 2 is...The surface of zone material is radiated in turn by waves of 10λ = 350nmand 540 nm respectively. The ratio of the s...In the Bohr model of a hydrogen atom, the centripetal force is furnished by the coulomb attraction between the proton an...