Ionic EquilibriumHard
Question
Choose the correct statement
Options
A.pH of acidic buffer solution decrease if more salt is added.
B.pH of acidic buffer solution increases if more salt is added.
C.pH of basic buffer decreases if more salt is added.
D.pH of basic buffer increases if more salt is added.
Solution
(B) is correct because pH = pKa + log 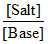 for acidic buffer.
for acidic buffer.
If [Salt] increases, pH of acidic buffer will increase.
pOH = pKb + log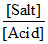 for basic buffer.
for basic buffer.
If [Salt] increases, pOH will increase, pH will decrease as pH = 14 - pOH.
Therefore (C) is correct but (D) is wrong.
(A) is not correct pH = pKa + log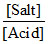
If [Salt] increase, pH will increase.
If [Salt] increases, pH of acidic buffer will increase.
pOH = pKb + log
If [Salt] increases, pOH will increase, pH will decrease as pH = 14 - pOH.
Therefore (C) is correct but (D) is wrong.
(A) is not correct pH = pKa + log
If [Salt] increase, pH will increase.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pOH of H2O is 7.0 at 298 K. If water is heated at 350 K, which of the following statement should be true?...The total number of different kind of buffers obtained during the titration of H3PO4 with NaOH are:...The following graph represents the titration of pH vs volume Find out the best possible statement from the graph shown....What is [H+] in mol/L of a solution that is 0.20 M in CH3COONa and 0.10 M in CH3COOH ? (Ka for CH3COOH = 1.8 × 10-5...The pH of 10-8 M solution of HCl in water is :...