Ionic EquilibriumHard
Question
What is [H+] in mol/L of a solution that is 0.20 M in CH3COONa and 0.10 M in CH3COOH ?
(Ka for CH3COOH = 1.8 × 10-5 )
(Ka for CH3COOH = 1.8 × 10-5 )
Options
A.3.5 × 10-4
B.1.1 × 10-5
C.1.8 × 10-5
D.9.0 × 10-6
Solution
CH3COOH (weak acid) and CH3COONa (conjugated salt) from acidic buffer and for acidic buffer,
pH = - log Ka + log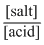 [∵ pKa = - log Ka]
[∵ pKa = - log Ka]
= - log (1.8 × 10-5) + log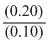
= 4.74 + log 2
= 4.74 + 0.3010 = 5.041
Now, [ H+ ] = antilog (- 5.045)
= 9.0 × 10-6 mol/L
pH = - log Ka + log
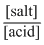 [∵ pKa = - log Ka]
[∵ pKa = - log Ka]= - log (1.8 × 10-5) + log
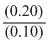
= 4.74 + log 2
= 4.74 + 0.3010 = 5.041
Now, [ H+ ] = antilog (- 5.045)
= 9.0 × 10-6 mol/L
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
What is the pH of 6.67 × 10−3 M aqueous solution of Al(OH)3 if its first dissociation is 100%, second dissociation is 50...The addition of sodium acetate to acetic acid solution will cause...Which is most stable ?...Which of the following statement(s) is/are correct?...How many grams of HCl should be dissolved in sufficient water to get 500 ml of an aqueous solution of pH, 2.0?...