Ionic EquilibriumHard
Question
pOH of H2O is 7.0 at 298 K. If water is heated at 350 K, which of the following statement should be true?
Options
A.pOH will decrease.
B.pOH will increase.
C.pOH will remain 7.0.
D.concentration of H+ ions will increase but that of OH- will decrease.
Solution
[OH-] = 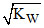 in pure water.
in pure water.
So as temperature increases Kw decreases ⇒ [OH-] decreases.
So as temperature increases Kw decreases ⇒ [OH-] decreases.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
As acidic buffer solution can be prepared by mixing the solutions of :...Ag+ + NH3 $\rightleftharpoons$ [Ag(NH3)+]; K1 = 1.6 × 103[Ag(NH3)+] + NH3 $\rightleftharpoons$ [Ag(NH3)2+]; K2 = 6.8 ×...The pH of 0. 1 M solution of the following salts increases in the order :...Which one of the following is NOT a buffer solution?...A volume of 20 ml of 0.8 M-HCN solution is mixed with 80 ml of 0.4 M-NaCN solution. Calculate the pH of the resulting so...