Ionic EquilibriumHard
Question
The following graph represents the titration of pH vs volume
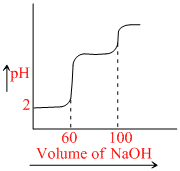

Find out the best possible statement from the graph shown.
Options
A.A diprotic acid.
B.Two monoprotic acids with the same Ka but different concentrations.
C.Two monoprotic acids with different Ka but the same concentration.
D.Two monoprotic acids with different Ka and different concentrations.
More Ionic Equilibrium Questions
Ratio of energy of photon of wavelength 3000 and 6000 is...A certain buffer solution contains equal concentration of X- and HX. The Kb for X- is 10-10. The pH of the buffer is:...Ionic mobility of which of the following alkali metal ions is lowest when aqueous solution of their salts are put under ...Let the solubility of an aqueous solution of Mg(OH)2 be x then its ksp is...1 M benzoic acid (pKa = 4.20) and 1M C6H5 COONa solutions are given separately. What is the volume of benzoic acid requi...