NEETIonic EquilibriumHard
Question
At what molar concentration of HCl will its aqueous solution have an [H+] to which equal contributions come from HCl and H2O.
Options
A.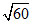 × 10-7 M
× 10-7 M
B.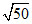 × 10-8 M
× 10-8 M
C.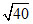 × 10-9 M
× 10-9 M
D.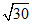 × 10-8 M
× 10-8 M
Solution
HCl → H+ + Cl-
(x + x) (x)
H2O ⇋ H+ + OH-
(x + x) (x)
Ionic product = Kw = (2x) (x) = 10-14
⇒ 2x2 = 10-14 ⇒ x =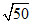 × 10-8
× 10-8
(x + x) (x)
H2O ⇋ H+ + OH-
(x + x) (x)
Ionic product = Kw = (2x) (x) = 10-14
⇒ 2x2 = 10-14 ⇒ x =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The volume of the water needed to dissolve 1 g of BaSO4 (KSP = 1.1 × 10-10) at 25oC is:...At 90o C, the hydronium ion concentration in pure water is 10−6 M. If 100 ml of 0.5 M-NaOH solution is mixed with 250 ml...Ksp of AgCl is 1.96 × 10-10. 100 mL of saturated AgCl solution is titrated with 1 × 10-5 M NH4 SCN. Volume of ...The solubility product of BaCrO4 is 2.4 × 10-10 M2. The maximum concentration of Ba(NO3)2 possible without precipit...Buffer solutions have constant acidity and alkalinity because:...