Ionic EquilibriumHard
Question
The volume of the water needed to dissolve 1 g of BaSO4 (KSP = 1.1 × 10-10) at 25oC is:
Options
A.280 litre
B.410 litre
C.205 litre
D.None of these
Solution
Solubility of BaSO4 = 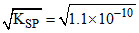 = 1.05 × 10-5 M
= 1.05 × 10-5 M
∴ wt. of BaSO4 = 1.05 × 10-5 × 233 = 244.37 × 10-5 g/litre.
∴ Volume of water needed to dissolve 1g BaSO4 is equal to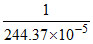 = 410 litre
= 410 litre
∴ wt. of BaSO4 = 1.05 × 10-5 × 233 = 244.37 × 10-5 g/litre.
∴ Volume of water needed to dissolve 1g BaSO4 is equal to
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The number of hydronium ions in 1 ml of an aqueous solution of pH 12.0 at 25o C is...Ca3(PO4)2 is insoluble in water. On adding a few drops of HCI to solid Ca3(PO4)2 in contact with water, the solid dissol...Four species are listed below i. HCO3- ii. H3O+ iii. HSO4- vi. HSO3F Which one of the following is the correct sequence ...The solubility product of a salt having general formula MX2, in water is: 4 × 10-12. The concentration of M2+ ions ...A volume of 250 ml of saturated clear solution of CaC2O4(aq) requires 6.0 ml of 0.001 M-KMnO4 in acidic medium for compl...