Ionic EquilibriumHard
Question
Ksp of AgCl is 1.96 × 10-10. 100 mL of saturated AgCl solution is titrated with 1 × 10-5 M NH4 SCN. Volume of 1 × 10-5 M NH4 SCN required precipitate all Ag+ from saturated AgCl solution as AgSCN is:
Options
A.140 mL
B.260 mL
C.70 mL
D.200 mL
Solution
m.moles of Ag+ in 100 ml of saturated solution of AgCl = 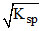 × 100 = 1.4 × 10-3
× 100 = 1.4 × 10-3
Now Ag+ + SCN- AgSCN (s)
m.moles of SCN- = m.moles of Ag+
1 × 10-5 × V = 1.4 × 10-3 ⇒ volume = 140 mL.
Now Ag+ + SCN- AgSCN (s)
m.moles of SCN- = m.moles of Ag+
1 × 10-5 × V = 1.4 × 10-3 ⇒ volume = 140 mL.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which of the following salts will give highest pH in water?...The degree of hydrolysis of a salt of weak acid and weak base in its 0.1 M solution is found to be 50%. If the molarity ...Solubility of BaF2 in a solution of Ba(NO3)2 will be represented by the concentration term...The solubility products of Al(OH)3 and Zn(OH)2 are 8.5 × 10-23 and 1.8 × 10-14 respectively. If NH4OH is added...An amount of 0.10 moles of AgCl(s) is added to one litre of water. Next, the crystals of NaBr are added until 75% of the...