Ionic EquilibriumHard
Question
Match list I (Solutions of salts of ...) with list II (pH of the solution is given by) and select the correct answer using the codes given below the lists:
List I List II
(a) Weak acid and strong base 1.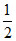 pKw
pKw
(b) Strong acid and weak base 2.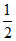 (pKw - pKb + pKa)
(pKw - pKb + pKa)
(c) Weak acid and weak base 3.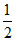 (pKw - pKb - log C)
(pKw - pKb - log C)
(d) Strong acid and strong base 4.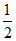 (pKw + pKa + log C)
(pKw + pKa + log C)
List I List II
(a) Weak acid and strong base 1.
(b) Strong acid and weak base 2.
(c) Weak acid and weak base 3.
(d) Strong acid and strong base 4.
Options
A.A → 1, B → 2, C → 3, D → 4
B.A → 4, B → 3, C → 2, D → 1
C.A → 4, B → 3, C → 1, D → 2
D.A → 3, B → 4, C → 2, D → 1
More Ionic Equilibrium Questions
M(OH)x has KSP 4 × 10-12 and solubility 10-4 M. The value of x is:...A + 2B ⇋ 2C + D, concentration of B was 1.5 time that of A, but the equilibriumconcentration of A and B are found ...1 M benzoic acid (pKa = 4.20) and 1M C6H5 COONa solutions are given separately. What is the volume of benzoic acid requi...Degree of hydrolysis for a salt of strong acid and weak base is...When 0.1 mole solid NaOH is added in 1lt of 0.1M NH3(aq) then which statement is going to wrong?(Kb = 2 × 10-5, log...