Ionic EquilibriumHard
Question
Degree of hydrolysis for a salt of strong acid and weak base is
Options
A.independent of dilution
B.increases with dilution
C.increases with decrease in Kb of the bases
D.decreases with decrease in temperature.
Solution
Let BA be this salt BA → B+ + A-
A- does not undergo hydrolysis because HA is strong acid. B+ undergoes hydroysis
h =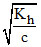 h = degree of hydrolysis where Kh = Hydrolysis constant =
h = degree of hydrolysis where Kh = Hydrolysis constant = 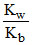
h ∝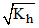 greater the hydrolysis constant greater the h(degree of hydrolysis).
greater the hydrolysis constant greater the h(degree of hydrolysis).
h ∝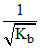 greater the Kb lesser the h.
greater the Kb lesser the h.
Hydrolysis is endothermic, Kh increases with temperature and h also increase with temperature.
h ∝ √ V = volume of salt solution hence h increases with dilution. Kh =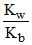
Both Kw and Kb change with temperature, hence Kh changes with temperature.
Hence, statement(B) is correct. h increases if Kb decreases, statement(C) correct.
It is found that as temperature increase, Kw and Kb increase but increase in Kw is greater than increase in Kb.
Hence, h increases with increase in temperature. or h decrease in temperature., hence statement.
(D) is correct.
(D) is not correct from explanation of (B).
A- does not undergo hydrolysis because HA is strong acid. B+ undergoes hydroysis
h =
h ∝
h ∝
Hydrolysis is endothermic, Kh increases with temperature and h also increase with temperature.
h ∝ √ V = volume of salt solution hence h increases with dilution. Kh =
Both Kw and Kb change with temperature, hence Kh changes with temperature.
Hence, statement(B) is correct. h increases if Kb decreases, statement(C) correct.
It is found that as temperature increase, Kw and Kb increase but increase in Kw is greater than increase in Kb.
Hence, h increases with increase in temperature. or h decrease in temperature., hence statement.
(D) is correct.
(D) is not correct from explanation of (B).
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
In the following reaction H2CO3 and C6H5NH2 respectively act as :-H2CO3 + C6H5NH2 ⇋ HCO3- + C6H5NH3+...Equimolar solutions of the following substances were prepared separately. Which one of these will record the highest pH ...pH of 0.01 M HS- will be:...One mole of CH3COOH and one mole of C2H5OH reacts to produce mol of CH3COOC2H2. The equilibrium constant is:...In which of the following cases, pH is greater than 7 ?...