Ionic EquilibriumHard
Question
When 0.1 mole solid NaOH is added in 1lt of 0.1M NH3(aq) then which statement is going to wrong?
(Kb = 2 × 10-5, log 2 = 0.3)
(Kb = 2 × 10-5, log 2 = 0.3)
Options
A.degree of dissociation of NH3 approaches to zero.
B.change in pH would be 1.85
C.conc of [Na+] = 0.1M, [NH3] = 0.1M, [OH-] = 0.2M
D.on addition of OH-, Kb of NH3 does not changes.
Solution
Initial pOH = 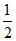 (pKb - log C) =
(pKb - log C) = 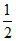 (4.7 - log 0.1) = 2.85
(4.7 - log 0.1) = 2.85
Final pOH = 1
Change in pOH = Change in pH = 1.85
Final pOH = 1
Change in pOH = Change in pH = 1.85
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The degree of hydrolysis of a salt of weak acid and weak base in its 0.1 M solution is found to be 50%. If the molarity ...A buffer solution is prepared in which the concentration of NH3 is 0.30 M and the concentration of NH4+ is 0.20 M. If th...The best indicator for the detection of end point in titration of a weak acid and a strong base is :...If ionization of XaYb takes place then, the number of Y–a ions will be equal to...At 30oC the solubility of Ag2CO3 (KSP = 8 × 10-12) would be greatest in one litre of:...