Ionic EquilibriumHard
Question
1 M benzoic acid (pKa = 4.20) and 1M C6H5 COONa solutions are given separately. What is the volume of benzoic acid required to prepare a 300 ml buffer solution of pH = 4.5 ? [log 2 = 0.3]
Options
A.200 ml
B.150 ml
C.100 ml
D.50 ml
Solution
pH = pKa + log 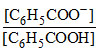 ⇒ 4.5 = 4.2 + log
⇒ 4.5 = 4.2 + log 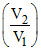 ⇒
⇒ 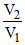 = 2
= 2
∴ volume of C6H5COONa required = V2 = 200 ml
volume of C6H5COOH required = V1 = 100 ml.
∴ volume of C6H5COONa required = V2 = 200 ml
volume of C6H5COOH required = V1 = 100 ml.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
In which of the following acid-base titration, pH is greater than 8 at equivalencepoint....Degree of hydrolysis for a salt of strong acid and weak base is...Which of the following is not correct :...Which shows the maximum rate of alkaline hydrolysis :-...Three solutions of strong electrolytes, 25 ml of 0.1 M-HX, 25 ml of 0.1 M-H2Y and 50 ml of 0.1 N-Z(OH)2 are mixed, the p...