Ionic EquilibriumHard
Question
KSP if Mg(OH)2 is 1 × 10-12. 0.01M MgCI2 will show precipitation in a solution of pH greater than:
Options
A.3
B.9
C.12
D.8
Solution
[Mg2+] [OH-]2 = 1 × 10-12 ;
∴ [OH-] =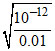 = 10-5
= 10-5
or pOH = 5.
and thus pH = 9.
∴ [OH-] =
or pOH = 5.
and thus pH = 9.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Silver ions are slowly added in a solution with [Br−] = [Cl−] = [CO32−] = [AsO43−] = 0.1 M. Which of the following compo...Unexposed silver halides are removed from photographic film when they react with sodium thiosulphate to form the complex...What % of the carbon in the H2CO3 - HCO3- buffer should be in the form of HCO3- so as to have a neutral solution? (Ka = ...The following graph represents the titration of pH vs volume Find out the best possible statement from the graph shown....50 mL of 0.1 M NaOH is added to 60 mL of 0.15 M H3PO4 solution (K1, K2 and K3 for H3PO4 are 10-3, 10-8 and 10-13 respect...