Ionic EquilibriumHard
Question
50 mL of 0.1 M NaOH is added to 60 mL of 0.15 M H3PO4 solution (K1, K2 and K3 for H3PO4 are 10-3, 10-8 and 10-13 respectively). The pH of the mixture would be about (log 2 = 0.3)
Options
A.3.1
B.5.5
C.4.1
D.6.5
Solution
H3PO4 + NaOH →NaH2PO4
9 5 0
4 0 5
pH = pKa + log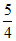 = 3 + 0.1 = 3.1
= 3 + 0.1 = 3.1
9 5 0
4 0 5
pH = pKa + log
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Assuming no change in volume, calculate the minimum mass of NaCl necessary to dissolve 0.01 mole of AgCl in 100 L soluti...At pair of R1 & R2 given tetrapeptide contain maximum positive charge at pH = 7...Which of the following is the strongest base :-...is converted in acetophenone by : -...Small amount of freshly precipitated magnesium hydroxides are stirred vigorously in a buffer solution containing 0.25 M ...