Ionic EquilibriumHard
Question
What % of the carbon in the H2CO3 - HCO3- buffer should be in the form of HCO3- so as to have a neutral solution? (Ka = 4 × 10-7)
Options
A.20 %
B.40 %
C.60 %
D.80%
Solution
pH = pKa + log 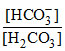 ⇒ 7 = 7 - log 4 + log
⇒ 7 = 7 - log 4 + log 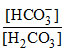 ⇒
⇒ 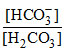 = 4
= 4
% of Carbon in the form of HCO3- =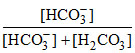 × 100 =
× 100 = 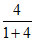 × 100 = 80 %
× 100 = 80 %
% of Carbon in the form of HCO3- =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The solubility of Fe(OH)3 would be maximum in...If pKb for fluoride ion at 25°C is 10.3, the ionization constant of hydrofluoric acid in water at this temperature is (l...Which of the following solutions when added to 1L of a 0.01 M CH3COOH solution will cause no change in the degree of dis...Which of the following will occur if a 0.1 M solution of a weak acid is diluted to 0.01 M at constant temperature?...In which of the following acid-base titration, pH is greater than 8 at equivalencepoint....