Ionic EquilibriumHard
Question
Precentage ionisation of weak acid can be calculated using the formula:
Options
A.100 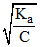
B.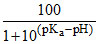
C.Both (A) and (B)
D.None of these
Solution
α = 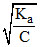 ∴ % α = 100
∴ % α = 100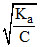 Also Ka =
Also Ka = 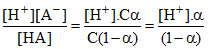
log Ka = logH+ + log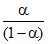 or pKα = pH + log
or pKα = pH + log 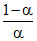
or pKa - pH = log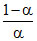 ∴
∴ 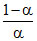 = 10pKa - pH
= 10pKa - pH
or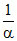 = 10pKa - pH + 1 or α =
= 10pKa - pH + 1 or α = 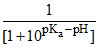
log Ka = logH+ + log
or pKa - pH = log
or
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Phenol on treatment with CO2 in the presence of NaOH followed by acidification produces compound X as the major product....50 mL of 2N acetic acid mixed with 10 mL of 1N sodium acetate solution will have an approximate pH of (Ka = 10-5)...The [OH-] in 100.0 ml of 0.016 M-HCl (aq) is :...Which solution will have pH closer to 1.0:...As acidic buffer solution can be prepared by mixing the solutions of :...