Ionic EquilibriumHard
Question
50 mL of 2N acetic acid mixed with 10 mL of 1N sodium acetate solution will have an approximate pH of
(Ka = 10-5)
(Ka = 10-5)
Options
A.4
B.5
C.6
D.7
Solution
Meq. of acetic acid = 50 × 2 = 100
Meq. of CH3COONa = 10 × 1 = 10
pH = - log Ka + log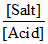 or pH = - log 10-5 + log
or pH = - log 10-5 + log 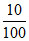 = 4
= 4
Meq. of CH3COONa = 10 × 1 = 10
pH = - log Ka + log
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Water in equilibrium with air contains 4.4 × 10−5% CO2. The resulting carbonic acid (H2CO3) gives the solution a hydroni...Which of the following compounds is not an antacid?...Aqueous saturated solution of sparingly soluble salt M2X (MW = 100) is observed to be 100 ppm then solubility product of...Approximate pH of 0.1 M aqueous H2S solution when K1 and K2 for H2S at 25oC are 1 × 10-7 and 1.3 × 10-13 respe...Concentration of the Ag+ ions in a saturated solution of Ag2C2O4 is 2.2 ×10-4 mol L-1 Solubility product of Ag2C2O4...