Ionic EquilibriumHard
Question
The [OH-] in 100.0 ml of 0.016 M-HCl (aq) is :
Options
A.5 × 1012 M
B.3 × 10-10 M
C.6.25 × 10-13 M
D.2.0 × 10-9 M.
Solution
[H+] = 0.016 M
[H+] [OH-] = 10-14 ⇒ [OH-] =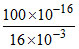 = 6.25 × 10-13 M
= 6.25 × 10-13 M
[H+] [OH-] = 10-14 ⇒ [OH-] =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Arsenic (III) sulphide form a sol with negative charge. Which of the following ionic substances will have lowest floceul...Which group of animals belong to the same phylum?...A definite volume of a N/20 CH3COOH (pKa = 4.7447) is titrated with a strong base (NaOH). It is found that 80 equal size...What is the pH value of M H2SO4 ?...KSP if Mg(OH)2 is 1 × 10-12. 0.01M MgCI2 will show precipitation in a solution of pH greater than:...