Ionic EquilibriumHard
Question
Which solution will have pH closer to 1.0:
Options
A.100 mL of (M/10) HCI + 100 mL of (M/10) NaOH
B.55 mL of (M/10) HCI + 45 mL of (M/10) NaOH
C.10 mL of (M/10) HCI + 90 mL of (M/10) NaOH
D.75 mL of (M/5) HCI + 25 mL of (M/5) NaOH
Solution
Meq. of HCI = 75 × 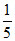 = 15 Meq. of NaOH = 25 ×
= 15 Meq. of NaOH = 25 × 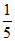 = 5
= 5
∴ Meq. of HCI left = 15 - 5 = 10 Now [HCI] left =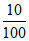 = 10-1
= 10-1
∴ pH = 1
∴ Meq. of HCI left = 15 - 5 = 10 Now [HCI] left =
∴ pH = 1
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
What will be the pH at the equivalence point during the titration of a 100 mL 0.2 M solution of CH3COONa with 0.2 M solu...The solubility of Ag2CO3 in water at 25oC is 1 × 10-4 mole/litre. What is its solubility in 0.01 M Na2CO3 solution?...Common hydrolysis so products of XeF2 & XeF4 are :-...Separate solutions of NaW, NaX, NaY and NaZ, each of concentrations 0.1 M has pH 7.0, 9.0, 10.0 and 11.0, respectively, ...The species present in solution when CO2 is dissolved in water is...