JEE Main | 2018Ionic EquilibriumHard
Question
Phenol on treatment with CO2 in the presence of NaOH followed by acidification produces compound X as the major product. X on treatment with (CH3CO)2O in the presence of catalytic amount of H2SO4 produces :
Options
A.
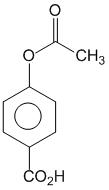
B.
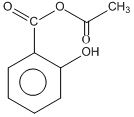
C.
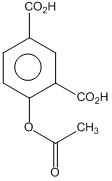
D.
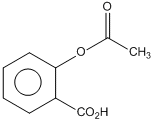
Solution
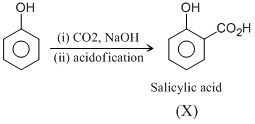
This is called Kolbe reactions.
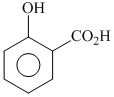 + (CH3CO)2O + conc H2SO4
+ (CH3CO)2O + conc H2SO4 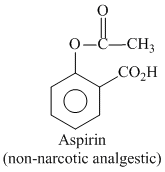
This step is called acylation reaction.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A cyclic process ABCD is shown in PV diagram for an ideal gas. Which of the following diagram represents the same proces...The pH of an aqueous solution of sodium chloride at 60o C is...Equal volumes of equimolar solutions of a weak acid and a weak base both having dissociation constant equal to 5 × ...The acid ionization constant of Zn2+ is 2.0 × 10−10. What is the basic dissociation constant of Zn(OH)+?...pH of a solution made by mixing 50 mL of 0.2 M NH4Cl and 75 mL of 0.1 M NaOH is :-[pKb of NH3(aq) = 4.74. log 3 = 0.47]...