Ionic EquilibriumHard
Question
A solution of Na2CO3 is added drop by drop to litre of a solution containing 10-4 mole of Ba2+ and 10-5 mole of Ag+, if KSP for BaCO3 is 8.1 × 10-9 and KSP for Ag2CO3 is 6.9 × 10-12 then which is not true:
Options
A.No precipitate of BaCO3 will appear until [CO32-] reaches 8.1 × 10-5 mol per litre
B.A precipitate of Ag2CO3 will appear when [CO32-] reaches 6.9 × 10-5 mol litre-1
C.No precipitate of Ag2CO3 will appear until [CO32-] reaches 6.9 × 10-2 mole per litre
D.BaCO3 will be precipitated first
Solution
For precipitation of Ag2CO3. [CO32-] = 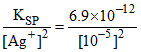 = 6.9 × 10-2
= 6.9 × 10-2
and for precipitation of BaCO3. [CO32-] =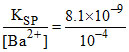 = 8.1 × 10-5
= 8.1 × 10-5
and for precipitation of BaCO3. [CO32-] =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A definite volume of a N/20 CH3COOH (pKa = 4.7447) is titrated with a strong base (NaOH). It is found that 80 equal size...The simultaneous solubilities of AgSCN and AgBr are, respectively (Ksp of AgSCN = 1 × 10–12, Ksp of AgBr = 2.1 × 10–13),...The pH of an acid buffer can be raised by 2 units by...Which solution will have pH closer to 1.0:...pH of 0.1M Na2HPO4 and 0.2M NaH2PO4 are respectively : pKa for H3PO4 are 2.2, 7.2 and 12.0....