Ionic EquilibriumHard
Question
pH of 0.1M Na2HPO4 and 0.2M NaH2PO4 are respectively : pKa for H3PO4 are 2.2, 7.2 and 12.0.
Options
A.4.7, 9.6
B.9.6, 4.7
C.4.7, 5.6
D.5.6, 4.7
Solution
For Na2HPO4, pH = 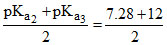 = 9.6.
= 9.6.
For Na2HPO4, pH =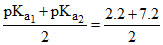 = 4.7.
= 4.7.
For Na2HPO4, pH =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A weak acid, HA is found to be 10% ionized in 0.01 M aqueous solution. Calculate thepH of a solution which is 0.1 M in H...When 20 mL of M/20 NaOH is added to 10 mL of M/10 HCl, the resulting solution will...The ratio of dissociation constant of two weak acids HA and HB is 4. At what moar concentration ratio, the two acids wil...For a sparingly soluble salt ApBq, the relationship of its solubility product (LS) with its solubility (S) is...When excess oxalic acid is added to CaCl2 solution, CaC2O4 is precipitated and the solution still contains some unprecip...