Ionic EquilibriumHard
Question
The pH of an acid buffer can be raised by 2 units by
Options
A.Increasing the concentration of both weak acid and salt by two units.
B.Increaseing the concentration of both the acid and salt by 10 times.
C.Diluting the solution by 10 times.
D.Increasing the concentration of the salt by 10 times and decreasing concentration of the acid by 10 times.
Solution
pH1 = pKa + log 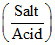
pH1 = pKa + log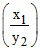
pH2 = pKa + log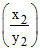
pH2 - pH1 = log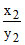 - log
- log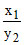
2 = log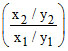
∴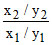 = 102 = 100
= 102 = 100
This is only possible, if the concentration of salt is increases by 10 times and the concentration of acid is decreased by 10 times
pH1 = pKa + log
pH2 = pKa + log
pH2 - pH1 = log
2 = log
∴
This is only possible, if the concentration of salt is increases by 10 times and the concentration of acid is decreased by 10 times
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pH at which a basic indicator with Kb = 1.0 × 10-10 changes colour when the concentration of indicator is 10-2 M :-...Complete hydrolysis of cellulose gives...The formation constant of Cu(NH3)42+ is 1.25 × 1012. What will be the equilibrium concentration of Cu2+ if 0.0125 moles ...The addition of ammonium chloride to acetic acid solution will cause...In which of the following solutions, the solubility of AgCN will be greater than that in pure water :Given Ksp(AgCN) = 4...