Ionic EquilibriumHard
Question
A definite volume of a N/20 CH3COOH (pKa = 4.7447) is titrated with a strong base (NaOH). It is found that 80 equal sized drops of NaOH, added from a burette effect the complete neutralisation. Find the pH, when the acid solution is neutralised to the extent of 20%.
Options
A.4.14
B.9.86
C.5.34
D.8.68
Solution
WA(CH3COOH) is titrated with SB(NaOH) and 20% of the WA is neutralised, so 20% of the salt of WA/SB (CH3COOH) is formed and 80% of the WA(CH3COOH) is left. So an acidic buffer is formed.
∴ [Acid] = 80; [Salt] = 20
pH = pKa + log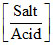
= 4.7447 + log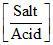
= 4.7447 + log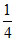
= 4.7447 - 2 log 2 = 4.7447 - 2 × 0.3010 = 4.14
∴ [Acid] = 80; [Salt] = 20
pH = pKa + log
= 4.7447 + log
= 4.7447 + log
= 4.7447 - 2 log 2 = 4.7447 - 2 × 0.3010 = 4.14
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The vapour pressure of a given liquid will decrease if....The solubility of Agl in Nal solutions is less than that in pure water because:...An acid type indicator, HIn differs in colour from its conjugate base (In–). The human eye is sensitive to colour differ...Phenol on treatment with CO2 in the presence of NaOH followed by acidification produces compound X as the major product....A volume of 250 ml of saturated clear solution of CaC2O4(aq) requires 6.0 ml of 0.001 M-KMnO4 in acidic medium for compl...