Gaseous StateHard
Question
An aqueous solution of X is added slowly to an aqueous solution of Y as shown in List-I. The variation in conductivity of these reactions is given in List-II. Match List-I with List-II and select the correct answer using the code given below the lists :
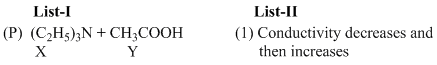
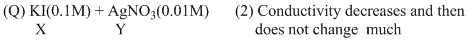
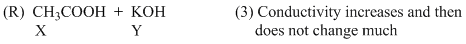
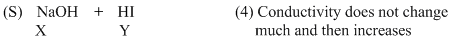
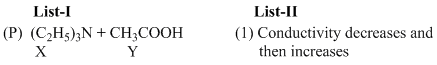
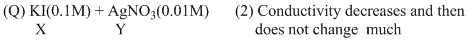
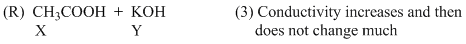
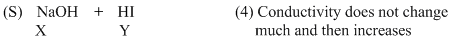
Options
A.P → 3, Q → 4, R → 2, S → 1
B.P → 4, Q → 3, R → 2, S → 1
C.P → 2, Q → 3, R → 4, S → 1
D.P → 1, Q → 4, R → 3, S → 2
Solution
(A) For weak base v/s weak acid titration upto equivalence point conductivity increases because weakelectrolyte (W.A.) is converted to strong electrolyte (salt) but after it weak base does not dissociateappreciably
(B) For KI + AgNO3 → AgI(ppt.) + KNO3
upto equivalence point number of ions remain same but after it increases
(C) For weak acid v/s strong base titration upto equivalence point OH- ion is replaced by CH3COO-so conductivity decreases but after it weak acid does not dissociate appreciably.
(D) For strong base v/s strong acid titration upto equivalence point H+ ion is replaced by Na+ so conductivitydecreases but after it increases due to increase in number of ions.
(B) For KI + AgNO3 → AgI(ppt.) + KNO3
upto equivalence point number of ions remain same but after it increases
(C) For weak acid v/s strong base titration upto equivalence point OH- ion is replaced by CH3COO-so conductivity decreases but after it weak acid does not dissociate appreciably.
(D) For strong base v/s strong acid titration upto equivalence point H+ ion is replaced by Na+ so conductivitydecreases but after it increases due to increase in number of ions.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
The compressibility factor for a real gas at highpressure is :-...2.5 L of a sample of a gas at 27oC and 1 bar pressure is compressed to a volume of 500 mL keeping the temperature consta...What volume will be occupied by a sample of gas at 40oC and 720 mm μg if its volume at 20oC and 750 mm μg was ...One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0L, 95K) → (4.0 atm, 5.0L, 245K) with a change...Which of following statement (s) is true I - Slope of isotherm at critical point is maximum. II - Larger is the value of...