Gaseous StateHard
Question
Which of following statement (s) is true
I - Slope of isotherm at critical point is maximum.
II - Larger is the value of TC easier is the liquification of gas.
III - Vander waals equation of state is applicable below critical temperature at all pressure.
I - Slope of isotherm at critical point is maximum.
II - Larger is the value of TC easier is the liquification of gas.
III - Vander waals equation of state is applicable below critical temperature at all pressure.
Options
A.only I
B.I & II
C.II & III
D.only II
Solution
I - Slope of isotherm below critical point < 0.
Slope of isotherm above critical point < 0.
Slope of isotherm at critical point = 0.
So slope of isotherm at critical point is maximum.
II- TC =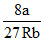
TC ∞ a
Larger value of TC It means less decreases in temperature is required to liquifly the gas. Gas will liquify at higher temperature. So, easier′ll be liquification.
III - When gas is below critical temperature. It is ′liquid′ so vander waal equation of state is not valid.
So, Answer (B).
Slope of isotherm above critical point < 0.
Slope of isotherm at critical point = 0.
So slope of isotherm at critical point is maximum.
II- TC =
TC ∞ a
Larger value of TC It means less decreases in temperature is required to liquifly the gas. Gas will liquify at higher temperature. So, easier′ll be liquification.
III - When gas is below critical temperature. It is ′liquid′ so vander waal equation of state is not valid.
So, Answer (B).
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
The average kinetic energy of an ideal gas per molecule in S.I. units at 25oC will be ?...Which of the following statement (s) is (are) correct ?...Kinetic theory of gases proves...What is the cordination number of sodium in Na2O ?...1g H2 gas at STP is expanded so that volume is doubled. Hence work done is :-...