Gaseous StateHard
Question
2.5 L of a sample of a gas at 27oC and 1 bar pressure is compressed to a volume of 500 mL keeping the temperature constant, the percentage increase in pressure is
Options
A.100 %
B.400 %
C.500%
D.80%
Solution
Using p1V1 = P2V2 1 × 2.5 = 0.5 × P2 = 5 bar.
∴ % increase in pressure =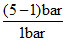 × 100% = 400 %.
× 100% = 400 %.
∴ % increase in pressure =
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
50 mL of each gas A and of gas B takes 200 and 150 s respectively for effusing through a pin hole under the similar cond...7.5 ml of a gaseous hydrocarbon was exploded with 36 ml of oxygen. The volume of gases on cooling was found to be 28.5 m...Schottky defect defines imperfection in the lattice structure of...The correction factor 'a' to the ideal gas equation corresponds to...The space lattice of graphite is...