Gaseous StateHard
Question
The compressibility factor for a real gas at highpressure is :-
Options
A.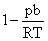
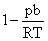
B.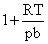
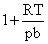
C.1
D.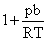
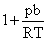
Solution
∴ For real gas, according to Vander wall′sequation
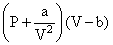 = RT (For 1 mole gas)
= RT (For 1 mole gas)
At high pressure
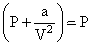
∴ P(V - b) = RT
PV = RT + Pb
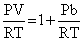
∴ Compressibility factor for a real gas
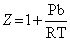
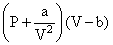 = RT (For 1 mole gas)
= RT (For 1 mole gas)At high pressure
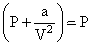
∴ P(V - b) = RT
PV = RT + Pb
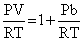
∴ Compressibility factor for a real gas
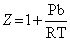
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
A substance AxBy crystallizes in a face-centred cubic (fcc) Lattice in which atoms ′A′ occupy each corner of...A real gas obeying Vander Waal′s equation will resemble ideal gas, if the :...On the surface of the earth at 1 atm pressure, a balloon filled with H2 gas occupies 500 mL. This volume is 5/6 of its m...One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0L, 95K) → (4.0 atm, 5.0L, 245K) with a change...The correct order of normal boiling points of O2, N2, NH3 and CH4, for whom the values of vander Waal′s constant &...