Gaseous StateHard
Question
On the surface of the earth at 1 atm pressure, a balloon filled with H2 gas occupies 500 mL. This volume is 5/6 of its maximum capacity. The balloon is left in air. It starts rising. The height above which the balloon will burst if temperature of the atmosphere remains constant and the pressure decreases 1 mm for every 100 cm rise of height is
Options
A.120 m
B.136.67 m
C.126.67 m
D.100 m
Solution
Max capacity of balloon = 600 ml
P1V1 = P2V2
500 × 1 = 600 × P2
P2 =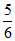 ×760 mm
×760 mm
= 633 mm
Height above which balloon will burst = (760 - 633) × 100 cm
= 127 × 100 cm
= 127 m
P1V1 = P2V2
500 × 1 = 600 × P2
P2 =
= 633 mm
Height above which balloon will burst = (760 - 633) × 100 cm
= 127 × 100 cm
= 127 m
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0L, 95K) → (4.0 atm, 5.0L, 245K) with a change...At what temperature will the total KE of 0.3 mol of He be the same as the total KE of 0.40 mol of Ar at 400 K ?...The ratio between the r.m.s. velocity of H2 at 50 K and that of O2 at 800 K is:...X ml of H2 gas effuses through a hole in a container in 5 sec. The time taken for the effusion of the same volume of the...At low pressures (For 1 mole), the Vander Waal′s equation is written as V = RTThe compressibility factor is then e...