Gaseous StateHard
Question
If Z is the number of atoms in the unit cell that represents the closest packing sequence..... ABC ABC ......, the number of tetrahedral voids in the unit cell is equal to :
Options
A.Z
B.2Z
C.Z/2
D.Z/4
Solution
In cubic closed pack system (CCP system).......ABCABC....... type of arrengement oflayers is found. In this system, there are atoms at the corners as well as center of the unit cell.
∴ No. of atoms per unit cell = 8 ×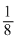 + 1 = 2
+ 1 = 2
Hence, the no. of tertrahedral voids in a unit cell = 2Z
∴ No. of atoms per unit cell = 8 ×
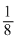 + 1 = 2
+ 1 = 2Hence, the no. of tertrahedral voids in a unit cell = 2Z
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Helium atom is two times heavier than a hydrogen molecule at 298 k, the average kinetic energy of helium is :...The pair(s) of ions where BOTH the ions are precipitated upon passing H2S gas in presence of dilute HCl, is(are)...A gaseous mixture containing He, CH4 & SO2 was allowed to effuse through a fine hole then find what molar ratio of gases...A gas cylinder containing cooking gas can withstand a pressure of 14.9 atmosphere. The pressure guaze of cylinder indica...The root mean square velocity of an ideal gas to constan pressure varies with density (d) as :...