Gaseous StateHard
Question
1 mol of an ideal gas at 27oC is compressed reverssibly from 20 L to 2L. Find out ᐃS :-
Options
A.4.606 cal / k
B.-4.606 cal / k
C.-19.147 J / k
D.Both (2) & (3)
Solution
ᐃS = 2.303 nR log 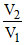
ᐃS = 2.303 × 1 × 8.31 log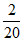
ᐃS = - 19.147 J/K or - 4.606 cal/k
ᐃS = 2.303 × 1 × 8.31 log
ᐃS = - 19.147 J/K or - 4.606 cal/k
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Calculate the entropy change involved in the isothermal reversible expansion of 5 mol of ideal gas from a volume of 10L ...The vander waal gas constant ′a′ is given by...Which is correctly matched :-...Consider given reaction : Metal (M) Select correct statement(s)...Critical constant for a real gas are given asTC = 180 K ; Vc = 0.1231 L/molePC = 45 atm ;The correct statement for the r...