Gaseous StateHard
Question
For one mole of a van der Waals gas when b = 0 and T = 300 K, the PV vs. 1/V plot is shown below. The value of the van der Waals constant a (atm.litre2mol-2) is
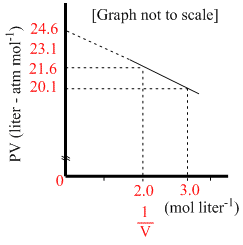

Options
A.1.0
B.4.5
C.1.5
D.3.0
Solution
van der Waals equation for 1 mole of real gas is,
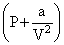 (V - b) = RT
(V - b) = RTbut, b = 0 (given)
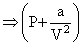 (V) = RT
(V) = RT∴ PV = - a ×
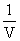 + RT .....(i)
+ RT .....(i)y = mx + c
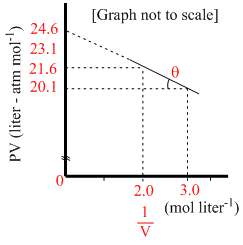
Slope = tan (π - θ) = - a
So, tan θ = a =
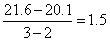
or tan θ =
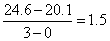
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
A substance AxBy crystallizes in a face-centred cubic (fcc) Lattice in which atoms ′A′ occupy each corner of...By what factor does the average velocity of a gaseous molecule increase when the temperature (in Kelvin) is doubled ?...At 100oC and 1 atm if the density of the liquid water is 1.0 g cm-3 and that of water vapour is 0.0006 g cm-3 then the v...Density of a Vander Waal′s gas at 500 K and 1 atm is found to be 0.8 kg/m3. It is also found that the gas diffuses...Which of the following does not shows explicitly the relationship between Boyle′s law and Charles′ law ?...