Chemical Kinetics and Nuclear ChemistryHard
Question
KI in acetone, undergoes SN2 reaction with each of P, Q, R and S. The rates of the reaction vary as -
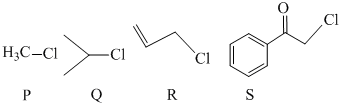

Options
A.P > Q > R > S
B.S > P > R > Q
C.P > R > Q > S
D.R > P > S > Q
Solution
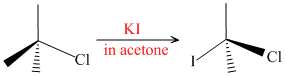 (SN2 Reaction)
(SN2 Reaction)Reaction takes place via formation of pentavalent C-transition state.
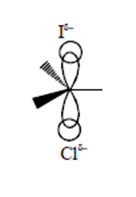
Transition state is stabilised by electron withdrawing group like
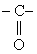 & not stabilised by electrondonating group like -CH3
& not stabilised by electrondonating group like -CH3So order of given compound for SN2 reaction
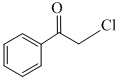 > CH3-Cl > CH2 = CH-CH2-Cl >
> CH3-Cl > CH2 = CH-CH2-Cl > 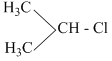
hence, Answer is (B)
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Among CaH2, BeH2, BaH2, the order of ionic character is...The increasing order of basicity of the following compounds is : (A)Â Â Â Â Â (B)Â Â (C)Â Â Â Â Â Â (D)Â...A(g) → B(g) + C(g) = k [A]At the start pressure is 100 mm and after 10 min , pressure is 120 mm. hence , rate cons...For the complex Ag+ + 2NH3 ⇋ [Ag(NH3)2+] = 2 × 107 L2 mol-2 s-1 [Ag+] [NH3]2 - 1 × 10-2 s-1 [Ag(NH3)2+]H...Select the correct statements out of I, II and III for zero order reactionI : Quantity of the product formed is directly...