JEE Main | 2018Chemical Kinetics and Nuclear ChemistryHard
Question
The increasing order of basicity of the following compounds is :
(A)Â Â  Â Â Â (B)Â Â
   (B)  
(C)Â Â 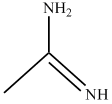 Â Â Â Â (D)Â
    (D) 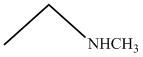
Options
A.
(b) < (a) < (c) < (d)
B.
(b) < (a) < (d) < (c)
C.
(d) < (b) < (a) < (c)
D.
(a) < (b) < (c) < (d)
Solution
Order of base nature depends on electron donation tendency.
In compound 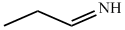 nitrogen is sp2 hybridized so least basic among all given compound.
nitrogen is sp2 hybridized so least basic among all given compound.
compound 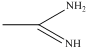 is very strong nitrogeneous organic base as lone pair of one nitrogen delocalize in resonance and make another nitrogen negativly charged and conjugate acid have two equivalent resonating structure.
is very strong nitrogeneous organic base as lone pair of one nitrogen delocalize in resonance and make another nitrogen negativly charged and conjugate acid have two equivalent resonating structure.
Thus it is most basic in given compouds.
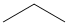 (secondary amine) more basic than
(secondary amine) more basic than
 (primary amine)
(primary amine)
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The radiations from a naturally occurring radioactive substance, as seen after deflection by a magnet in one direction, ...For the following reaction(CH3)3CCl + H2O → (CH3)3 COH + HCl, = k[CH3)3CCl], hence, rate determining step can be:...For the non-equilibrium process, A + B → products, the rate is first-order w.r.t. A and second-order w.r.t. B. If ...The rate constant for the reaction, 2N2O5 → 4NO2 + O2 is 3.0 × 10-4s-1. If start made with 1.0 mol L-1 of N2O...For a reaction A 2B starting with 1 M of ′A′ only, concentration of B (in M) after 100 sec. and 200 sec. is ...