Chemical Kinetics and Nuclear ChemistryHard
Question
Select the correct statements out of I, II and III for zero order reaction
I : Quantity of the product formed is directly proportional to time
II : Larger the initial concentration of the reactant, greater the half-life period
III : If 50% reaction takes place in 100 minutes, 75% reaction take place in 150 minutes.
I : Quantity of the product formed is directly proportional to time
II : Larger the initial concentration of the reactant, greater the half-life period
III : If 50% reaction takes place in 100 minutes, 75% reaction take place in 150 minutes.
Options
A.I only
B.I and II only
C.II and III only
D.I, II and III
Solution
We know for zeroth order Rxn
For (I) x = kt
For (II) t1/2 ∞ [A0] [Initial concentration]
For (III) A0 -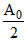 = k × 100 .....(i)
= k × 100 .....(i)
A0 -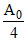 = k × t .....(ii)
= k × t .....(ii)
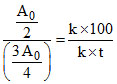
t =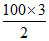 = 150 minutes.
= 150 minutes.
For (I) x = kt
For (II) t1/2 ∞ [A0] [Initial concentration]
For (III) A0 -
A0 -
t =
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
At 325°C, 1,3-butadiene dimerizes according to the equation: 2C4H6(g) → C8H12(g). It is found that the partial pressure ...Which of the following pair of substances illustrates law of multiple proportions :-...The physical adsorption of gases on the solid surface is due to:...The rate constant for the reaction, 2N2O5 → 4NO2 + O2 is 3.0 × 10-4s-1. If start made with 1.0 mol L-1 of N2O...A certain zero order reaction has k = 0.025 M s-1 for the disappearance of A. What will be the concentration of A after ...