Chemical Kinetics and Nuclear ChemistryHard
Question
For the complex Ag+ + 2NH3 ⇋ [Ag(NH3)2+]
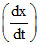 = 2 × 107 L2 mol-2 s-1 [Ag+] [NH3]2 - 1 × 10-2 s-1 [Ag(NH3)2+]
= 2 × 107 L2 mol-2 s-1 [Ag+] [NH3]2 - 1 × 10-2 s-1 [Ag(NH3)2+]
Hence, ratio of rate constants of the forward and backward reactions is :
Hence, ratio of rate constants of the forward and backward reactions is :
Options
A.2 × 107 L2 mol-2
B.2 × 109 L2 mol-2
C.1 × 10-2 L2 mol-2
D.0.5 × 10-9 L2 mol-2
Solution
Equation : Ag+ + 2NH3 ⇋ [Ag(NH3)2+]
Given,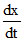 = 2 × 107 L2 mol-2 s-1 [Ag+] [NH3]2 - 1 × 10-2 s-1 [Ag(NH3)2+]
= 2 × 107 L2 mol-2 s-1 [Ag+] [NH3]2 - 1 × 10-2 s-1 [Ag(NH3)2+]
For equilibrium
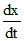 = 0
= 0
2 × 107 [Ag+] [NH3]2 -1 × 10-2 [Ag(NH3)2+] = 0
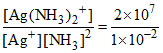 = 2 × 109 L2 mol-2.
= 2 × 109 L2 mol-2.
Given,
For equilibrium
2 × 107 [Ag+] [NH3]2 -1 × 10-2 [Ag(NH3)2+] = 0
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Decrease in atomic number is observed during :...As the initial concentration increases from 0.75 to 1.55 M in a reaction, t1/2 decreases from 60 to 29 s. The order of t...t1/4 can be taken as the time taken for the concentration of a reactant to drop to of its initial value. If the rate con...Which of the following statement is incorrect about half-life period :...Which of the following is not a green house gas?...